Lectin Chaperone System: The Glycoprotein Binding Proteins 
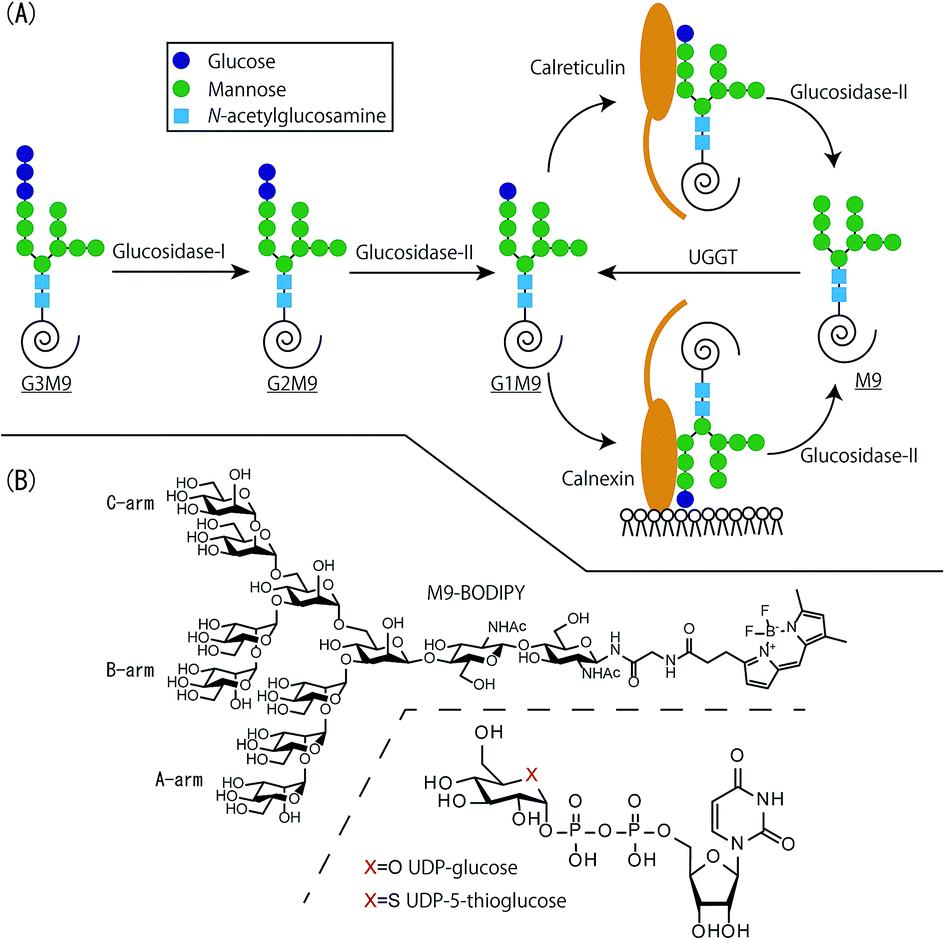
In case the calnexin system in the lumen fails to salvage a misfolded protein, it is transported to the cytoplasm for degradation. The primary players of this quality check pipeline are the lectins, ER-resident chaperones, and a glucosyl transferase enzyme. The calnexin cycle is a specific chaperone system that folds and assesses the confirmation of N-glycosylated proteins before they can exit the ER lumen. The reglucosylation recycles the polypeptide for another round of folding by calnexin/calreticulin chaperones until the correct protein structure is achieved.ĮR is the primary site for the maturation and folding of soluble and transmembrane secretory proteins. Otherwise, UGGT1 adds a glucose molecule from UDP-glucose back to the glycan attached to the polypeptide. If the folding is correct, the protein is allowed to exit the ER. The UDP-glucose:glycoprotein glucosyltransferase 1, or UGGT1 enzyme, then assesses the protein for its folding accuracy. Soon after, glucosidase II trims out the final glucose residue, creating non-glucosylated glycans on the polypeptide. The lectins then recruit other chaperones to assist in polypeptide folding. The membrane-bound calnexin and soluble calreticulin are lectin chaperones, which specifically bind such monoglucosylated trimmed glycans. Two of these glucose residues are sequentially removed by the action of glucosidase I and glucosidase II, leaving behind a trimmed N-linked oligosaccharide with a single glucose. 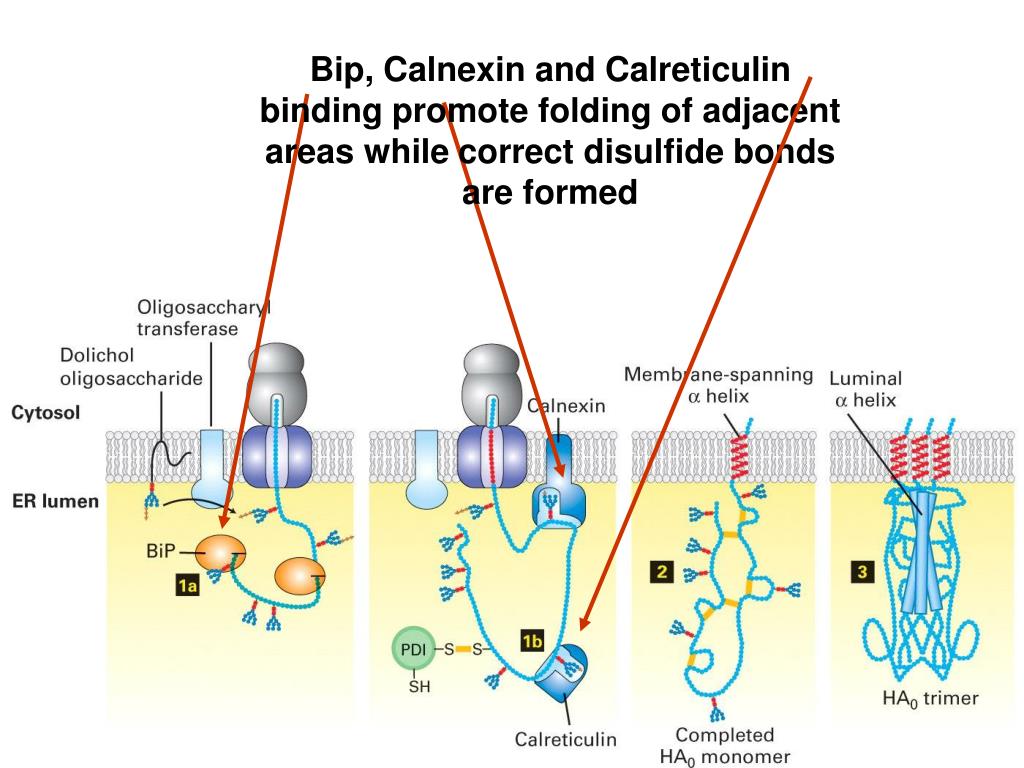
These precursor glycans are branched oligosaccharides, each with three terminal glucose residues. In the ER lumen, an unfolded polypeptide has multiple precursor N-linked glycans attached to it.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
